Vitamin C Content
For two experiments the vitamin C content of orange juice was assessed using spectroscopy. This experiment allowed us to see the effect that different treatments had on the nutrient content of orange juice. The two methods used were Spectroscopy and HPLC
Protocol Spectroscopy:
A standard curve was first generated using the following procedure:
Four concentrations of ascorbic acid were created by diluting a 200 microgram/mL stock solution. The stock solutions were then reacted with the indicator DCPIP and the absorbance was taken 520 nm.
Protocol Spectroscopy:
A standard curve was first generated using the following procedure:
Four concentrations of ascorbic acid were created by diluting a 200 microgram/mL stock solution. The stock solutions were then reacted with the indicator DCPIP and the absorbance was taken 520 nm.
Results: After the standard curve was generated we determined the vitamin c content by reacting it with DCPIP and determining a dilution that will give us a value that lay on the curve
Sample E Boiled orange juice: 1/30 dilution
Absorbance: .328
Concentration: 4.304 µg/ml
Adjusted concentration: 129.12 µg/ml
Sample B chilled orange juice: 1/18 dilution
Absorbance: .326
Concentration: 4.304 µg/ml
Adjusted concentration: 77.47 µg/ml
Sample D Room Temperature orange juice: 1/36 dilution
Absorbance: .265
Concentration: 4.304 µg/ml
Adjusted concentration: 154.95 µg/ml
For this lab we had some difficulty getting results that were within our standard curve and had to change the DCPIP used in the redox reactions multiple times. Our results may be inconsistent due to our reaction conditions changing throughout this experiment.
Sample E Boiled orange juice: 1/30 dilution
Absorbance: .328
Concentration: 4.304 µg/ml
Adjusted concentration: 129.12 µg/ml
Sample B chilled orange juice: 1/18 dilution
Absorbance: .326
Concentration: 4.304 µg/ml
Adjusted concentration: 77.47 µg/ml
Sample D Room Temperature orange juice: 1/36 dilution
Absorbance: .265
Concentration: 4.304 µg/ml
Adjusted concentration: 154.95 µg/ml
For this lab we had some difficulty getting results that were within our standard curve and had to change the DCPIP used in the redox reactions multiple times. Our results may be inconsistent due to our reaction conditions changing throughout this experiment.
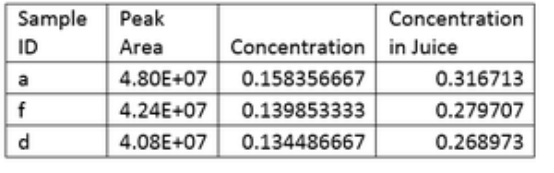
Protocol HPLC:
In this lab we determined the content of three types of orange juice samples by performing HPLC. We first generated a standard curve with samples of known concentrations of vitamin c. After the standard curve was generated, we took 1:2 dilutions of samples of orange juice stored under three different conditions. In this experiment reverse phase chromatography was used and the mobile phase was 2% K3PO4 and a C18 column was used.
In this lab we determined the content of three types of orange juice samples by performing HPLC. We first generated a standard curve with samples of known concentrations of vitamin c. After the standard curve was generated, we took 1:2 dilutions of samples of orange juice stored under three different conditions. In this experiment reverse phase chromatography was used and the mobile phase was 2% K3PO4 and a C18 column was used.
For this experiment the results were more consistent with our expectations because the experiment conditions did not have to change with each of the samples.