Bomb Calorimetry
A bomb calorimeter is an instrument used to measure the
calorific value (heat of combustion) of a sample. Calories
are the heat energy required to heat one gram of water
to one degree Celsius.
calorific value (heat of combustion) of a sample. Calories
are the heat energy required to heat one gram of water
to one degree Celsius.
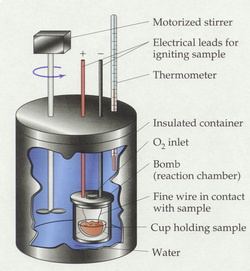
Bomb Calorimetry Components:
- Crucible
- Wire-String
- Steel Chamber
- Jacket
- Supply of Oxygen
- Stirrer
- Water
- Thermometer

Bomb Calorimetry Protocol
- Completely grind the sample and condense the 0.5-1.0 gram sample into a pellet.
- Place the pellet on the dried crucible and twist a string from the ignition wire on the bomb.The string should be close or on the pellet.
- Fasten the bomb inside and begin the run.
Results:
- Veggie Burger: 5.52 kcal/g --> 186.9 kcal Nutrition facts: 110 kcal per serving
- Spinach Dip: 6.52 kcal/g --> 50.2 kcal Nutrition facts: 50 kcal per serving
- Pickled Herring: 3.26 kcal/g --> 162.9 kcal Nutrition facts: 120 kcal per serving
- Dried Seaweed: 7.88 kcal/g --> 141.8 kcal Nutrition facts: 70 kcal per serving
